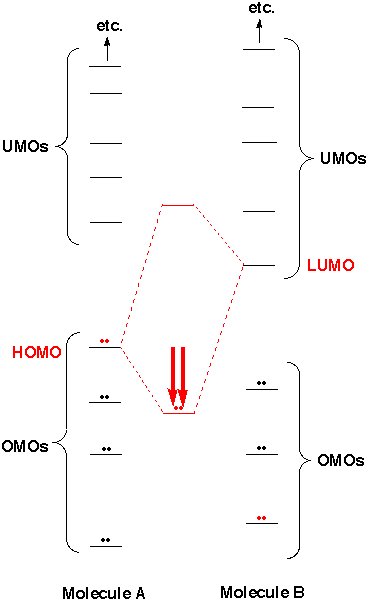
For example, molecular orbitals are artifacts in WFT, whereas in DFT, electron density plays the dominant role. Wave function theory (WFT) and density functional theory (DFT)-the two most popular solutions to electronic structure problems of atoms and molecules-share the same origin, dealing with the same subject yet using distinct methodologies. Huang, Ying Rong, Chunying Zhang, Ruiqin Liu, Shubin If no, then simply move on, and try other descriptors (atomic charges/spin populations/bond orders at transition states, for example) instead - there wasn't a strong reason for you to expect a good correlation in the first place.Evaluating frontier orbital energy and HOMO/LUMO gap with descriptors from density functional reactivity theory. If yes, you can comment that the correlation exists, or that the trend of the HOMO-LUMO gaps contributes to the trend of the reactivities, but unless you provide evidence that the aforementioned four approximations hold, you should not say that you "explained" the reactivities by the HOMO-LUMO gaps. Thus my suggestion is: observe if your HOMO-LUMO gap data do correlate with your reactivities. by exploiting an accidental cancellation of the contributions of $\langle \Psi_0 | V | \Psi_0 \rangle$, $|\langle \Psi_0 | V | \Psi_I \rangle|$, the $O(V^3)$ term, and the exciton binding energy. 
I said never, because even if the HOMO-LUMO gap does correlate perfectly with the reactivity, you are achieving this correlation largely by luck, i.e. If any of the conditions are violated, then you can never explain your reactivity based on the HOMO-LUMO gap. \Delta E & = \langle \Psi_0 | V | \Psi_0 \rangle - \sum_$ reflects the trend of $\omega_1$ and finally $|\langle \Psi_0 | V | \Psi_1 \rangle|$ must be roughly constant among the molecules to be studied. Then from second-order perturbation theory, the (ground state) energy change caused by the perturbation is Suppose that the ground state wavefunction of the molecule is $|\Psi_0\rangle$, the Hamiltonian of the molecules is $H$, and the perturbation is expressed as an operator $V$. These two cases can be unified as: a reactive molecule is one that tends to significantly lower its energy in the presence of a perturbation, where the perturbation can be either external (from other molecules) or internal (from the structural change of the molecule itself). A molecule is said to be reactive, if either (1) it reacts exothermically and quickly with many kinds of molecules, or (2) it decomposes exothermically and quickly by itself. The correlation between the HOMO-LUMO gap and reactivity stems from second-order perturbation theory. As a result, more and more people try to use the HOMO-LUMO gap to rationalize their reactivity patterns, while not realizing that this does not work nearly as well as they thought. In addition, among those people who ever attempted to compare the HOMO-LUMO gap against the reactivity, usually only those people who do find a correlation will publish their results, leading to considerable survivorship bias. Many sources, however, only point out the correlation between the HOMO-LUMO gap and reactivity, but fail to point out that the correlation only holds in the statistical sense, and is by no means universal. The HOMO-LUMO gap is only weakly correlated with the reactivity of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
